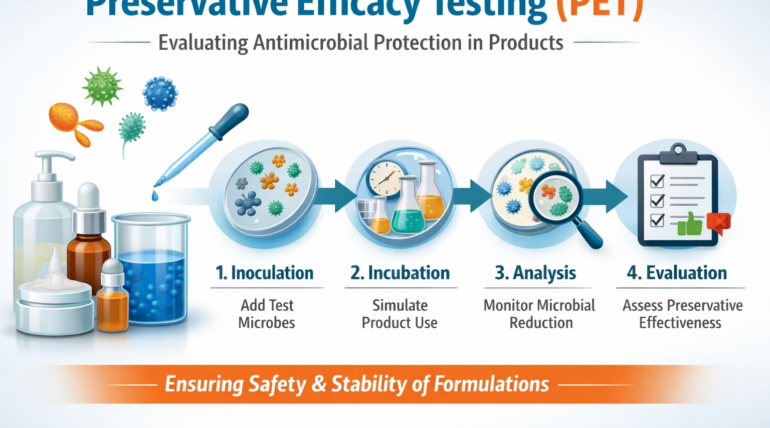
What Is Preservative Efficacy Testing (PET)?
Preservative efficacy testing (PET), also known as preservative challenge testing, is used to evaluate how effectively a product’s preservative system protects against microbial growth over time. PET assesses whether a formulation can control bacteria, yeast, and mold under conditions that simulate contamination during normal use.
This type of testing is commonly applied to cosmetics, personal care products, and other water-containing formulations where microbial growth may present safety, quality, or regulatory concerns.
Why Preservative Efficacy Testing Is Important
Products that contain water or moisture-supporting ingredients are inherently susceptible to microbial contamination. During manufacturing, packaging, storage, and consumer use, products may be exposed to microorganisms that can compromise product safety and stability.
Preservative efficacy testing helps:
- Demonstrate that a preservative system is effective
- Reduce risk of microbial spoilage
- Support product safety assessments
- Provide data for regulatory and quality documentation
PET is often a critical component of product development, reformulation, and ongoing quality programs.
What Preservative Efficacy Testing Evaluates
During preservative efficacy testing, a product is intentionally inoculated with specific microorganisms. The ability of the preservative system to reduce and control microbial levels is then monitored over defined time intervals.
Typical evaluations focus on:
- Reduction of microbial populations
- Control of regrowth over time
- Overall robustness of the preservative system
The results help determine whether a formulation provides adequate protection throughout its intended shelf life and use period.
Common Microorganisms Used in PET
While specific organisms may vary by standard or product type, PET commonly evaluates effectiveness against representative:
- Bacteria
- Yeast
- Mold
These organisms are selected to challenge the preservative system under controlled but rigorous conditions.
PET Testing Standards and Guidelines
Preservative efficacy testing is typically conducted in accordance with recognized international or pharmacopeial standards. Common references include:
- ISO 11930 – Cosmetics: Evaluation of antimicrobial protection
- USP <51> – Antimicrobial effectiveness testing
Different standards may define acceptance criteria, test organisms, and evaluation timelines differently. Selection of the appropriate standard depends on product category, market, and regulatory expectations.
Preservative efficacy testing is commonly performed in alignment with recognized standards such as ISO 11930 and USP <51>, which outline challenge testing principles and acceptance criteria.
Preservative Efficacy Testing vs. Microbial Limits Testing
Although both tests relate to microbiological quality, they serve different purposes:
- Microbial limits testing evaluates the level of microorganisms present in a product at a specific point in time.
- Preservative efficacy testing evaluates whether the preservative system can control microbial growth over time following contamination.
PET is a dynamic challenge test, while microbial limits testing is a snapshot assessment.
When Is Preservative Efficacy Testing Performed?
PET may be performed:
- During product development or formulation screening
- After reformulation or preservative changes
- Prior to product launch
- When extending shelf life or use claims
- As part of ongoing quality or risk-management programs
Testing is often repeated when formulation, packaging, or manufacturing conditions change.
Limitations of Preservative Efficacy Testing
While PET provides valuable insight into preservative performance, it does not guarantee absolute sterility or predict all real-world contamination scenarios. Results should be interpreted alongside formulation design, packaging selection, and manufacturing controls.
PET is best viewed as one component of a broader product safety and quality strategy.
Summary
Preservative efficacy testing is an essential tool for evaluating whether a product’s preservative system can effectively control microbial growth throughout its intended use. By challenging formulations under standardized conditions, PET helps support product safety, quality, and regulatory compliance.
For products that rely on preservatives to maintain microbiological integrity, PET plays a key role in risk reduction and product stewardship.
Preservative Effectiveness / Challenge Testing – Pentyl Labs

