Shop
Description
Preservative Effectiveness Testing (PET) – Ph. Eur. 5.1.3
Preservative effectiveness testing evaluates how well a preservative system controls microbial growth in a product over time. Pentyl Labs performs this testing in accordance with European Pharmacopoeia (Ph. Eur.) 5.1.3 – Efficacy of Antimicrobial Preservation. The method supports product safety, stability, and regulatory compliance.
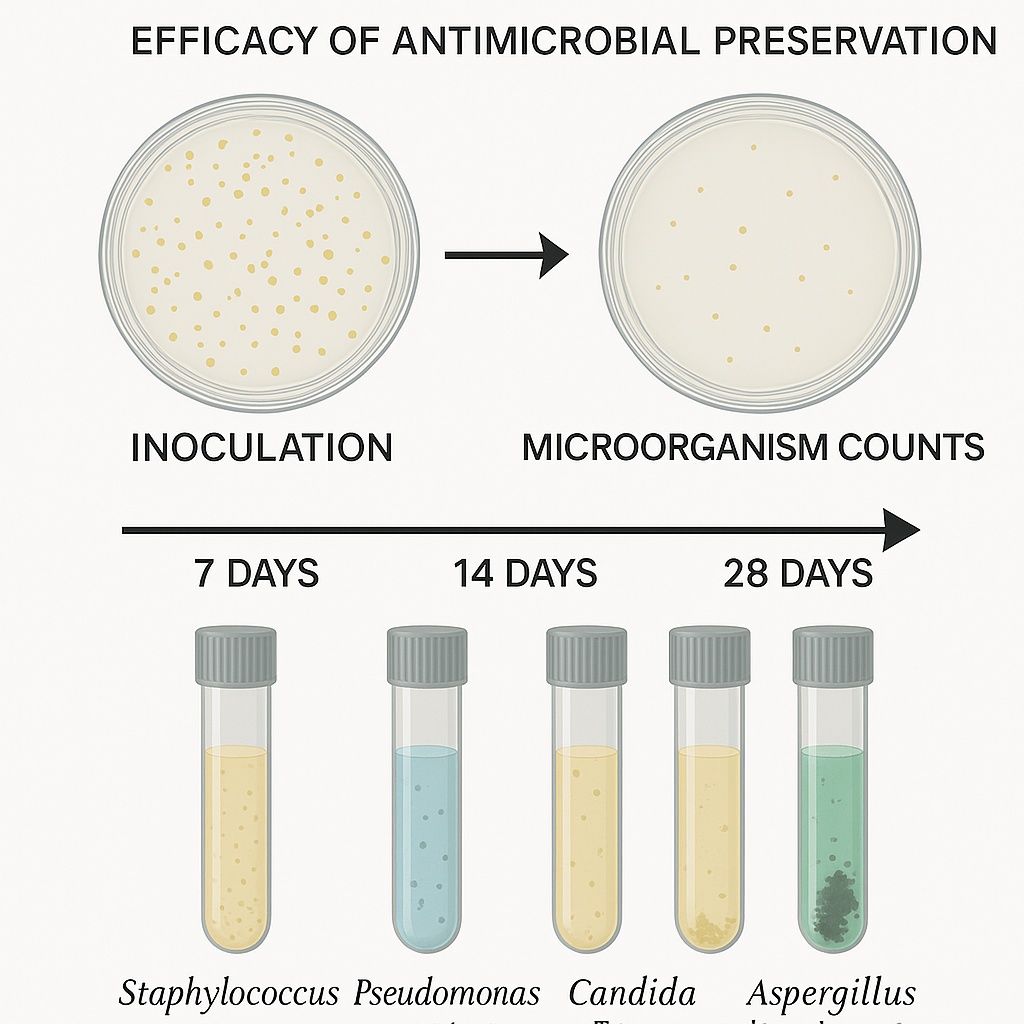
During preservative effectiveness testing, analysts intentionally inoculate the product with standardized challenge microorganisms. These organisms include Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, Candida albicans, and Aspergillus brasiliensis. Together, they represent common bacterial and fungal contaminants encountered during manufacturing and consumer use.
After inoculation, analysts store the product under controlled conditions and sample it at defined time points. Typical evaluation points include Day 7, Day 14, and Day 28. At each interval, analysts quantify surviving microorganisms using validated recovery methods. Results show log reduction or growth inhibition relative to the initial inoculum.
Preservative effectiveness testing applies different acceptance criteria depending on product category. These categories include parenteral, ophthalmic, topical, and oral products. Category A represents the most stringent antimicrobial performance requirements. Analysts compare results against the applicable criteria to determine compliance.
Pentyl Labs includes validated neutralization procedures to ensure accurate microbial recovery. The laboratory also applies quantitative plate counting and statistical review to confirm preservative performance. This approach provides reliable and reproducible data.
Manufacturers often request preservative effectiveness testing during formulation development, preservative selection, reformulation, or prior to market launch. The data supports regulatory submissions, product information files, and internal quality programs. Because many regions restrict animal testing, PET using standardized microbiological methods has become the accepted industry standard.
This service is commonly included within broader Preservative Efficacy Testing and Microbiological Testing Services programs. Official guidance from the European Pharmacopoeia Commission defines test design, organisms, and acceptance criteria.
Overall, preservative effectiveness testing provides clear evidence that a product’s preservative system performs as intended and meets regulatory expectations.