Shop
Description
Introduction
The Formulation Health Score™ helps brands understand how cosmetic and personal care formulations perform during accelerated stability testing. Pentyl Labs developed the Formulation Health Score™ to convert complex stability data into a clear evaluation of formulation performance.
Traditional stability studies often record individual observations at specific timepoints. However, these measurements alone do not always explain how formulation ingredients interact over time. Therefore, the Formulation Health Score™ evaluates several stability indicators together rather than reviewing them separately.
During a stability program, analysts examine formulation behavior under accelerated storage conditions, typically around 40 °C. As a result, potential degradation pathways and packaging interactions become visible sooner. Consequently, product developers can identify possible stability challenges earlier in the development process.
How the Formulation Health Score™ Works
First, Pentyl Labs collects stability data from structured stability studies. These studies typically include several timepoints under controlled accelerated conditions. Next, analysts evaluate multiple formulation indicators such as physical stability signals, pH drift, microbial trends, and packaging interaction observations.
Additionally, the evaluation focuses on how these indicators change over time. For example, gradual pH drift combined with microbial growth may indicate preservative system stress. Similarly, visible color changes together with packaging interaction signals may suggest compatibility concerns.
Because the Formulation Health Score™ considers several indicators together, it provides a broader understanding of formulation performance. Therefore, development teams can identify risks sooner and make better decisions about formulation improvements or additional testing.
Data Used to Calculate the Formulation Health Score™
Several indicators contribute to the final evaluation.
Stability Behavior
Analysts monitor physical stability indicators such as:
-
color change
-
phase separation
-
sediment formation
-
odor changes
-
viscosity variation
These observations help identify trends across stability timepoints.
pH Drift
Analysts also measure pH during the stability study. These measurements help identify:
-
initial pH conditions
-
pH drift over time
-
compatibility with preservative systems
Consequently, pH monitoring helps reveal early signs of chemical instability.
Microbial Trends
When microbiological testing is included, analysts review microbial results such as:
-
total aerobic counts
-
yeast and mold results
-
pathogen screening
Together, these results provide insight into preservative system performance over time.
Packaging Interaction Risk
Packaging compatibility observations may include:
-
container discoloration
-
odor migration
-
leakage or swelling
-
dispensing issues
In many cases, packaging interactions can influence long-term product stability.
Formulation Health Score™ Methodology
Pentyl Labs calculates the Formulation Health Score™ using a weighted evaluation model that considers several indicators of formulation stability.
| Evaluation Category | Weight |
|---|---|
| Stability Behavior Signals | 35% |
| Microbial Trend | 20% |
| pH Drift | 15% |
| Preservative Robustness | 15% |
| Packaging Interaction Risk | 15% |
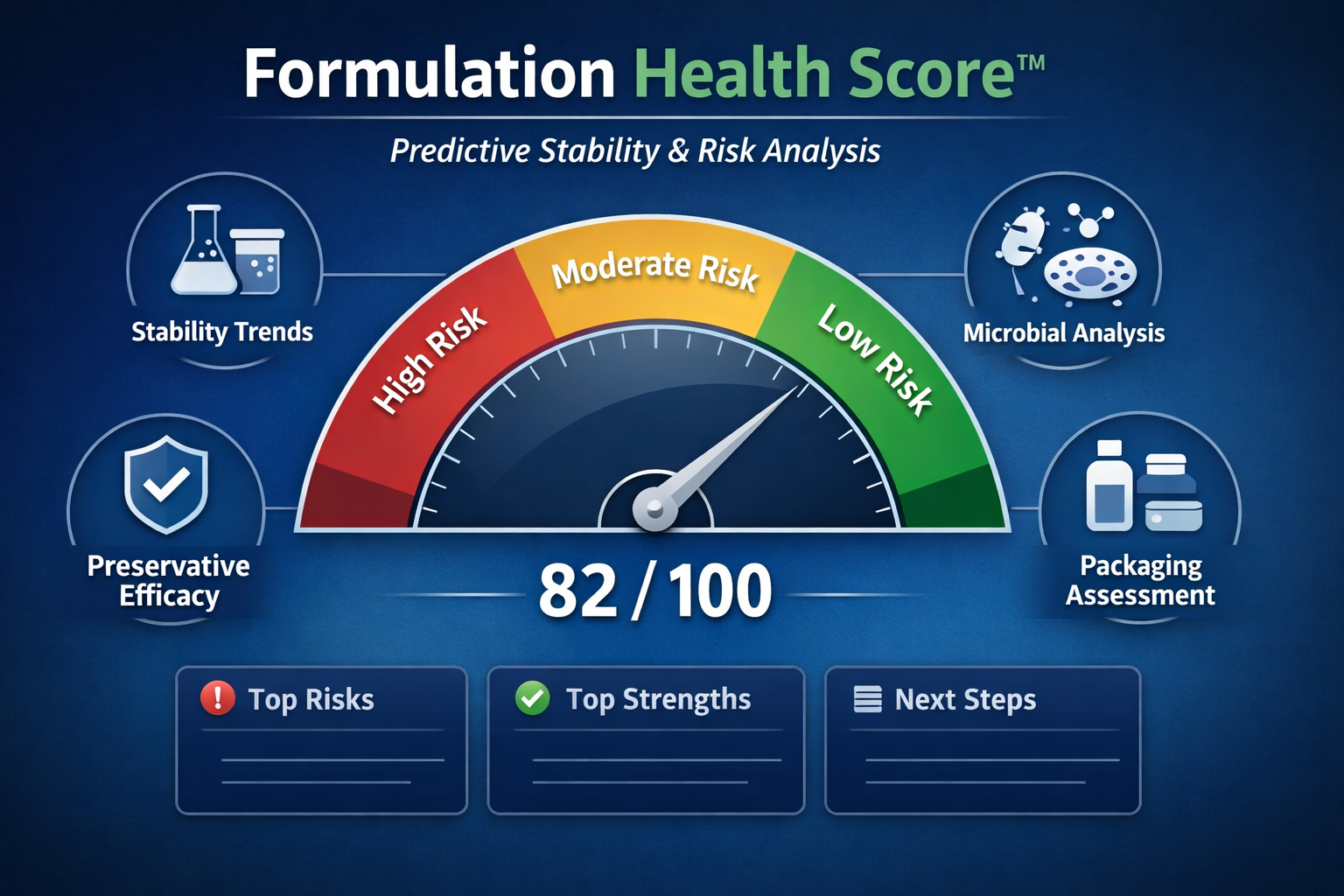
The evaluation produces a Formulation Health Score™ ranging from 0 to 100, which represents the overall stability profile of the formulation during the study period.
Interpreting the Formulation Health Score™
| Score Range | Interpretation |
|---|---|
| 85–100 | Strong formulation stability |
| 70–84 | Stable with moderate monitoring recommended |
| 50–69 | Elevated formulation risk |
| Below 50 | Significant instability risk identified |
In addition to the numerical score, the report includes interpretation of stability trends and recommendations for formulation optimization or additional testing.
What the Formulation Health Score™ Report Includes
Each Formulation Health Score™ report may include:
-
overall formulation health score (0–100)
-
category score breakdown
-
interpretation of stability trends
-
identification of formulation risks
-
identification of formulation strengths
-
recommended next steps for formulation development
This structured approach helps translate complex stability data into practical insight for formulation scientists and product development teams.
Related Pentyl Labs Services
The Formulation Health Score™ may be performed alongside additional Pentyl Labs testing services.
Stability Testing
https://www.pentyllabs.com/all-services/stability-testing/
Microbiological Testing
https://www.pentyllabs.com/microbiological-testing/
Packaging Compatibility Testing
https://www.pentyllabs.com/packaging-compatibility-testing/
Regulatory Context
Cosmetic stability testing supports product safety and regulatory readiness. In the United States, cosmetic safety requirements fall under the Modernization of Cosmetics Regulation Act (MoCRA).
More information is available here:
https://www.fda.gov/cosmetics/cosmetics-laws-regulations/modernization-cosmetics-regulation-act-2022-mocra
International cosmetic stability guidance is also available through the International Organization for Standardization (ISO):
https://www.iso.org/standard/75058.html