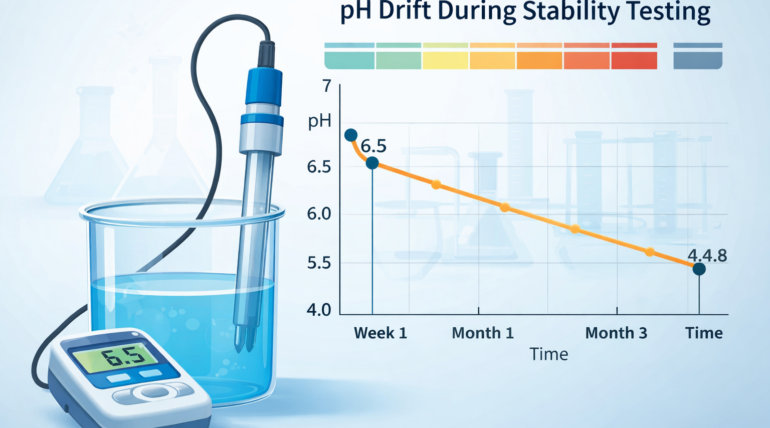
pH drift during stability testing refers to the change in pH that occurs in a formulation over time under controlled storage conditions. Monitoring pH throughout stability studies is essential because shifts in acidity or alkalinity can indicate chemical degradation, preservative instability, ingredient incompatibility, or packaging interactions.
For cosmetics, personal care products, and household formulations, even small pH changes may affect product safety, performance, preservative efficacy, and regulatory compliance. As a result, pH monitoring is a standard component of most stability testing programs.
Why pH Drift Occurs During Stability Testing
Several mechanisms can cause pH drift during a stability study. Understanding these mechanisms helps formulators identify potential formulation weaknesses early in product development.
Chemical Degradation
Many ingredients gradually degrade when exposed to heat, oxygen, or light. Degradation reactions often produce acidic or basic byproducts that alter formulation pH.
Examples include:
- Hydrolysis of esters
- Oxidation reactions
- Decomposition of natural extracts
- Fragrance component breakdown
Accelerated stability testing conditions (such as 40°C or elevated humidity) can accelerate these reactions.
Preservative System Changes
Preservatives are often pH-dependent. Over time, interactions with other ingredients may alter the formulation pH, reducing preservative effectiveness.
For example:
- Organic acid preservatives require acidic environments
- Some preservative salts hydrolyze over time
- Microbial metabolism can also alter pH if contamination occurs
pH monitoring during stability testing helps ensure the preservative system remains within its effective operating range.
Ingredient Interactions
Formulations contain complex mixtures of surfactants, emulsifiers, oils, and active ingredients. Over time these materials may interact and alter the acid-base balance.
Common examples include:
- Neutralization reactions
- Buffer depletion
- Emulsion breakdown releasing acidic fatty acids
- Reaction of actives with formulation bases
These interactions may not be immediately visible but can appear as gradual pH drift during stability testing.
Packaging Interactions
Containers and packaging components may also influence pH stability.
Potential mechanisms include:
- Migration of additives from packaging materials
- Absorption of acidic or basic ingredients
- Permeation of oxygen or moisture
These effects are often evaluated during packaging compatibility and stability studies.
Acceptable pH Change During Stability Testing
There is no universal regulatory limit for acceptable pH drift. However, industry guidance and internal specifications typically define acceptable ranges.
Common guidelines include:
| Product Type | Typical Acceptable pH Change |
|---|---|
| Cosmetics & personal care | ±0.3 – ±0.5 pH units |
| Preservative-sensitive formulations | ±0.2 – ±0.3 |
| Buffered formulations | Minimal change expected |
If pH drift exceeds the defined specification, formulators may need to investigate:
- ingredient degradation
- preservative failure
- packaging incompatibility
- manufacturing variability
Why pH Monitoring Is Critical in Stability Studies
pH measurement is one of the most sensitive indicators of formulation instability. Even when products appear physically stable, pH drift can signal early formulation breakdown.
Monitoring pH during stability testing helps detect:
- preservative system failure
- chemical degradation
- emulsion instability
- packaging interactions
- microbial contamination risks
Because pH influences skin compatibility, product performance, and microbiological stability, maintaining consistent pH throughout shelf life is essential.
When pH Is Measured During Stability Testing
During stability programs, pH is typically measured at defined time intervals.
Typical testing schedule:
| Timepoint | Purpose |
|---|---|
| Initial | Baseline measurement |
| 1 month | Early instability detection |
| 3 months | Accelerated stability trend |
| 6 months | Shelf-life projection |
| 12 months | Real-time stability confirmation |
Measurements are usually performed using calibrated benchtop pH meters in accordance with ASTM E70 or equivalent laboratory procedures.
Best Practices for Preventing pH Drift
Formulators can reduce the risk of pH drift by incorporating several stability-focused design strategies.
Best practices include:
- using appropriate buffer systems
- selecting pH-stable preservatives
- verifying ingredient compatibility
- evaluating packaging compatibility
- performing accelerated stability studies early
These steps help ensure products maintain consistent pH throughout their intended shelf life.
Stability Testing Services for Cosmetic Formulations
Monitoring pH drift is a standard part of cosmetic stability programs. Stability studies evaluate physical, chemical, and functional changes in formulations under controlled conditions.
Typical parameters evaluated include:
- pH
- viscosity
- color
- odor
- separation
- packaging compatibility
Pentyl Labs performs cosmetic and consumer product stability testing programs designed to identify formulation changes such as pH drift before products reach the market.
Conclusion
pH drift during stability testing provides valuable insight into the long-term behavior of cosmetic and consumer formulations. Tracking pH over time helps identify degradation pathways, preservative risks, and formulation incompatibilities that could impact product safety or performance.
By incorporating pH monitoring into stability studies, manufacturers can ensure products remain safe, stable, and effective throughout their intended shelf life.

