Shop
Description
50-Subject HRIPT for Sensitive Skin
This 50-subject human repeat insult patch test evaluates the irritation and sensitization potential of cosmetic and personal care products using a panel composed specifically of individuals with sensitive skin. The study supports product safety reviews, claim substantiation, and retailer or regulatory documentation needs.
Because products intended for sensitive or reactive skin require additional scrutiny, this study design focuses on a higher-risk population. As a result, the data generated is more relevant for products marketed toward sensitive skin consumers.
What This Study Evaluates
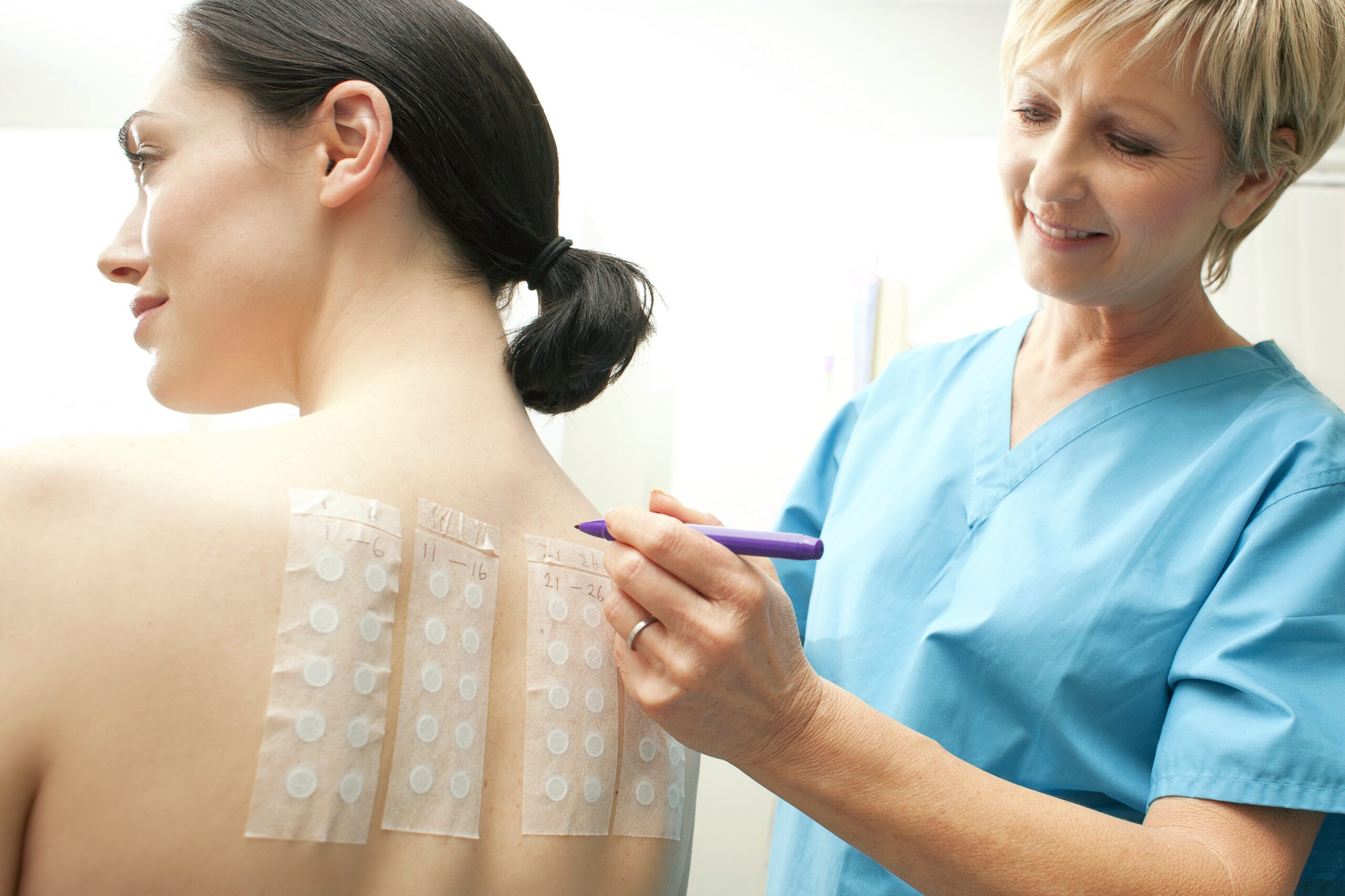
A Human Repeat Insult Patch Test (HRIPT) is a controlled clinical study used to assess whether repeated topical exposure to a product may cause irritation or allergic sensitization. In this study, qualified sensitive-skin panelists receive repeated patch applications under occlusive conditions, followed by a rest period and challenge phase.
Throughout the study, trained clinical personnel monitor skin responses at each exposure. Therefore, both short-term irritation and delayed sensitization responses can be evaluated under standardized conditions.
Clinical patch testing is commonly conducted under dermatologist oversight and aligns with accepted dermatological testing practices supported by organizations such as the American Academy of Dermatology.
What the Study Includes
This 50-subject sensitive-skin panel study typically includes:
-
Recruitment of qualified sensitive-skin participants
-
Repeated patch applications under controlled conditions
-
Scheduled skin evaluations during induction and challenge phases
-
Medical oversight and documentation of observations
-
A final report summarizing irritation and sensitization outcomes
In addition, study protocols follow established clinical testing practices to ensure consistency and reliable interpretation of results.
Claims Commonly Supported
Data from this type of study may support claims such as:
-
Suitable for sensitive skin
-
Dermatologist tested
-
Non-irritating
-
Hypoallergenic (when supported by results and proper context)
Because claim acceptability varies by retailer and market, results should always be reviewed in conjunction with marketing and regulatory guidance.
Products Commonly Evaluated
This study is commonly requested for:
-
Skin care and facial products
-
Leave-on cosmetic formulations
-
Baby and pediatric products
-
Products marketed for sensitive or reactive skin
Why Choose Pentyl Labs?
Pentyl Labs coordinates clinical skin testing studies with clear protocols, transparent timelines, and practical reporting. Moreover, we support related services such as safety assessments, label reviews, and claim support. As a result, study data integrates smoothly into broader product development and compliance workflows.
📩 Contact [email protected] to request a 50-Subject HRIPT for Sensitive Skin.