Shop
Description
50 Subject HRIPT Testing – Normal Skin
Overview of the Study
50 Subject HRIPT testing evaluates whether a cosmetic or personal care product may cause skin irritation or sensitization after repeated use. This study helps brands confirm product safety and supports both regulatory and marketing needs.
Importantly, many regions restrict or ban animal testing for cosmetics. Because of these restrictions, companies rely on well-established human clinical studies to assess skin compatibility. As a result, HRIPT has become a widely accepted standard for evaluating irritation and sensitization in finished products.
Study Design and Panel Details
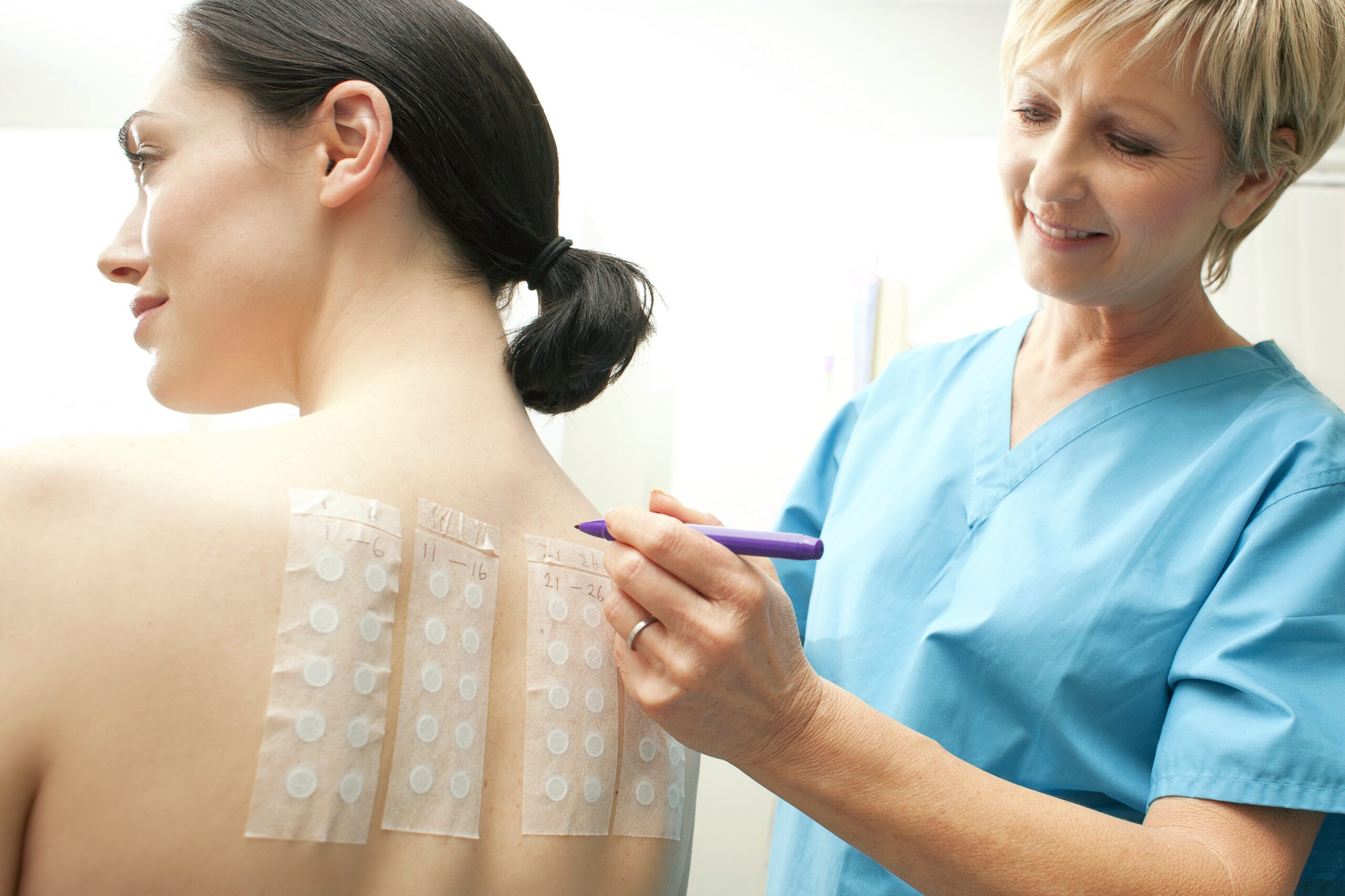
The Human Repeat Insult Patch Test uses a panel of 50 healthy adult participants with normal skin. Trained clinical staff apply the test product to the skin following a defined application schedule. Panelists receive repeated applications during the induction phase, which allows clinicians to observe skin responses over time.
After a rest period, the study includes a challenge application. This final step helps identify any delayed reactions or signs of sensitization. Throughout the study, clinical staff document all observations in a controlled and consistent manner.
Medical Oversight and Reporting
A board-certified dermatologist supervises the study from start to finish. The dermatologist reviews skin responses, evaluates clinical findings, and signs the final report. This medical oversight strengthens the reliability of the data and supports responsible use in safety reviews and marketing materials.
Following study completion, the final report summarizes observations, outcomes, and overall skin compatibility. Sponsors can use this documentation for internal reviews, regulatory support, and claim development.
Claims Supported by HRIPT Testing
Because animal testing is restricted in many markets, 50 Subject HRIPT testing serves as a key method for supporting common cosmetic claims, including:
-
Dermatologist tested
-
Clinically tested
-
Hypoallergenic
-
Non-irritating
Before the study begins, sponsors define claim language. This step ensures the protocol aligns with marketing goals while remaining accurate and defensible.
Optional Pediatric Review and Program Integration
For products intended for children or sensitive populations, an optional pediatrician signature is available upon request. This added review may help address retailer, regulatory, or internal requirements.
Many brands include this study within broader Safety and Compliance Testing and Claim Substantiation Testing programs. In addition, the U.S. Food and Drug Administration (FDA) encourages manufacturers to support cosmetic claims with appropriate scientific data derived from human-relevant testing methods.
Why Brands Use This Study
Overall, 50 Subject HRIPT testing provides a reliable and ethical approach to evaluating repeated skin exposure. Because animal testing is no longer permitted in many regions, this study has become a trusted industry standard for reducing product risk and strengthening consumer confidence before launch.